Introduction
If you’re looking for a cheap dual battery setup for your Ford Ranger or Bronco II, this guide shows you one of the simplest and most affordable ways to do it using a battery isolator solenoid and basic wiring.
A dual battery system allows you to run accessories like lights, radios, air compressors, or camping gear without draining your main starting battery. This setup uses a continuous-duty solenoid (relay) that automatically connects both batteries when the engine is running and separates them when the key is off—so you’ll always have enough power to start your truck.
This is a budget-friendly dual battery system that doesn’t require an expensive DC-DC charger. It’s ideal for off-road use, overlanding, or anyone who needs extra battery capacity without a complicated install.
Important: This setup is designed for older charging systems and will NOT work properly on 2019 and newer Ford Ranger models due to their smart alternator systems. For those trucks, you’ll need a DC-DC charger-based setup instead.
Automotive Battery Basics: Learn how a car battery works, how to test it, how to charge it, and how to choose the right battery for your vehicle. This guide covers voltage, cold cranking amps (CCA), battery types, charging methods, maintenance, and common battery problems.
An automotive battery is a 12-volt lead-acid battery that stores electrical energy and supplies power to start your engine and run electrical components. A fully charged battery should read about 12.6 volts with the engine off, and 13.6–14.5 volts while the engine is running.
Quick Answers
- What voltage should a car battery be? 12.6V (engine off), 13.6–14.5V (running)
- What does CCA mean? Cold Cranking Amps — the battery’s ability to start in cold weather
- When is a battery considered dead? Around 12.0 volts or lower
- How long do car batteries last? Typically 3–5 years
Note: This article is on batteries themselves and not on adding multiple batteries. It also does not troubleshoot charging systems.
Battery Safety
Always wear proper eye, face and hand protection when working with battery
Never lean over battery while boosting, testing, or charging
Exercise caution when working with metallic tools or conductors to prevent short circuits and arcing
Keep terminals protected to prevent accidental shorting
Replace any battery that has signs of damage to the terminals, case, or cover
Install battery in a ventilated area for operation and during charging
If your battery states to not add water to it, do not add water.
How It Works
The modern automotive battery is a lead-acid storage design. In short, it’s an electrochemical device that converts chemical energy into electrical energy. When the battery is placed under a load, such as when the ignition is switched on, the device converts stored chemicals into electricity, and the current flows through the wires to its destination.
A standard 12-volt, lead-acid battery is made up of six cells connected in series. Each cell produces approximately 2.1 volts.
2.1 volts x 6 = 12.6 volts
The plates are formed into a plate group, which holds a number of plates of the same polarity (for instance, all positive or all negative). The like-charged plates are welded to a post strap. The plate groups are then alternated within the battery — positive, negative, positive, negative. There is usually one extra set of negative plates to balance the charge. To ensure that the different plate groups don’t touch each other, non-conductive sheets called separators are inserted between them.
The plates are made up of lead with separate plates of lead oxide, which are submerged into an electrolyte solution of about 35% sulfuric acid and 65% water. The mixture of acid and water should have a specific gravity of 1.265 at 80 degrees Fahrenheit. Specific gravity refers to the weight of a solution, with water having a reference rating of 1.000. The electrolyte in an automotive battery is therefore just slightly over one-and-a-quarter times the weight of regular water.
A chemical reaction releases electrons, allowing them to flow through conductors to produce electricity. As the battery discharges, the acid of the electrolyte reacts with the materials of the plates, changing their surface to lead sulfate. When the battery is recharged, the chemical reaction is reversed: the lead sulfate reforms into lead oxide and lead. With the plates restored to their original condition, the process may now be repeated.
Electrolyte Solution is 65% water + 35% sulfuric acid
Charge (How Much Power’s In The Battery?)
As mentioned above, A standard 12-volt, lead-acid battery is made up of six cells connected in series. Each cell produces approximately 2.1 volts.
2.1 volts x 6 = 12.6 volts
A car battery is considered charged at 12.4 volts or higher. It is considered discharged when it’s at 12.39 volts or less. When a battery drops voltage, even a small amount, it makes a big difference. For instance, when a battery drops from 12.6 to 12.0 volts, its power drops from 100% to 25%. At 12.4 volts, a car battery is 75% charged. At 12.2 volts, it’s 50% charged.
Measuring The Voltage
With the battery charged, open circuit voltage should be 12.6 to 12.8 volts with the engine off and all loads off. With the car running and all loads off, the voltage should be between 13.6 and 14.5 volts.
Disable the engine by pulling the fuel pump fuse. Measure the battery voltage while cranking the engine. It should read at least 9.5 to 10 volts while cranking.
If the charging voltage is low, suspect some charging system problem. But if the charging voltage is correct and the cranking voltage is low, then the battery is suspect, as you’ve already troubleshot the wiring from the battery to the starter and ground.
Some manufacturers include a built-in hydrometer to show the state of charge of the battery. This Lucite “eye” has a float immersed in the electrolyte. When the battery is charged, the specific gravity of the electrolyte increases, and the colored top of the float is visible in the window. When the battery is discharged (or if the electrolyte level is too low), the float sinks and the window appears yellow (or black). The built-in hydrometer only checks the state of charge of one cell and will not show faults in the other cells. In a non-sealed battery each of the cells can be checked with a portable hydrometer. Batteries will last longer if not stored in the discharged state. In emergencies a battery can be jump started, by the battery of another vehicle or by a hand portable battery booster (jump pack).
Charging System
In normal automotive service the vehicle’s engine-driven alternator powers the vehicle’s electrical systems and restores charge used from the battery during engine cranking. When your engine is running, there should be 13-15 volts at the battery terminals.
When installing a new battery or recharging a battery that has been accidentally discharged completely, one of several different methods can be used to charge it. The most gentle of these is called trickle charging. Other methods include slow-charging and quick-charging, the latter being the harshest.
Battery Charging
Both fast- and slow-charging units are used to recharge batteries. Each has its advantages. Fast chargers are the most popular. They charge batteries at a higher rate or charge–usually 40 amperes for 12-volt batteries and 70 amperes for 6-volt batteries. At this rate, fast chargers can recharge most batteries in about 1 hour. However, batteries must be in good condition to accept a fast charge. Sulfation on the plates of the battery can lead to excessive gassing, boiling, and heat buildup during fast charging. Never fast charge a battery that shows evidence of sulfation buildup or separator damage.
Trickle chargers provide low charging currents of about 5 to 15 amperes. Slow charging may require 12 to 24 hours but is the only safe way of charging sulfated batteries. In general, almost any battery can be charged at any current rate as long as excessive electrolyte gassing does not occur and the electrolyte temperature does not exceed 125°F (51.6°C). However, when time is available, slow charging is the safest and easiest method to use. In fact, many fast chargers can be adjusted to provide slow charging. The basic rule of thumb for slow charging a battery is 1 ampere for each positive plate in one cell. Use the chart below to determine the rate of charge according to the reserve capacity of the battery.
Perform charging in a well-ventilated area away from sparks and open flames.
Always be sure the charger is off before connecting or disconnecting the leads to the battery.
Remember to wear eye protection, and never attempt to charge a frozen battery.
All battery chargers have manufacturer-specific characteristics and operating instructions that must be followed.
When charging a battery in the vehicle, always disconnect the battery cables to avoid damaging the generator or other electrical components.
Maintaining A Charge During Storage – Check below about storing your battery for more charging information.
WARNING: DO NOT exceed the manufacturer’s battery charging limits. Also, never charge the battery if the built-in hydrometer registers clear or light yellow. Replace the battery.
Jump Starting
When it is necessary to jump-start a car with a discharged battery using a booster battery and jumper cables, follow the instructions below to avoid damaging the charging system or creating a hazardous situation. Always wear eye protection when making or breaking jumper cable connections.
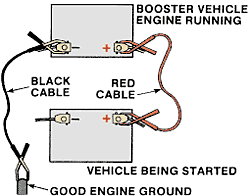
Proper setup and connections for jump-starting a vehicle with a low battery.
The following steps should be followed to safely jump-start most vehicles:
1) Make sure the two vehicles are not touching each other. The excessive current flow through the vehicle’s bodies can damage the small ground straps that attach the engine block to the frame. These small wires are designed to carry only 30 amperes. If the vehicles are touching, as much as 400 amperes may be carried through them.
2) For each vehicle, engage the parking brake and put the transmission in neutral or park.
3) Turn off the ignition switch and all accessories, on both vehicles.
4) Attach one end of the positive jumper cable to the disabled battery’s positive terminal.
5) Connect the other end of the positive jumper cable to the booster battery’s positive terminal.
6) Attach one end of the negative jumper cable to the booster battery’s negative terminal.
Another option to jump starting the vehicle is a jump pack. It’s a lot more convenient since you don’t need a second vehicle to jump start the vehicle. Jump packs plug in to a household outlet and can even be charged with an optional cord that plugs in to a cigarette lighter/DC jack.

Types of Batteries
Lead-acid batteries for automotive use are made with slightly different construction techniques, depending on the application of the battery. The typical battery in use today is of the “flooded cell” type, indicating liquid electrolyte. AGM or Absorbed Glass Mat type batteries have no free liquid electrolyte and are gaining acceptance by consumers in SLI (Starting – Lighting – Ignition) applications. This article deals with the flooded type of car battery.
Starting Battery – The starting (cranking) or shallow cycle type is designed to deliver large bursts of energy, usually to start an engine. The SLI batteries usually have a greater plate count in order to have a larger surface area that provides high electric current for short period of time. The plates are good for lots of amps, briefly. But they break down under deep cycling–meaning lots of current drawn out of them. They should not be drawn down to under 90% capacity. Once the engine is started, they are recharged by the engine driven charging system. If you draw large amounts of current from your battery such as from winching and draw the batteries down under 90%, you could be building up damage in the battery.
Deep Cycle – The deep cycle (or motive) type is designed to continuously provide power for long periods of time (for example in a trolling motor for a small boat, auxiliary power for a recreational vehicle, or traction power for a golf cart or other battery electric vehicle). They can also be used to store energy from a photo voltaic array or a small wind turbine. They usually have thicker plates in order to have a greater capacity and survive a higher number of charge/discharge cycles. They but can be drawn down to 50% regularly without damage. The energy to weight ratio, or specific energy, is in the range of 30 Wh/kg (108 kJ/kg).
Some battery manufacturers claim their batteries are dual purpose (starting and deep cycling).
Maintenance Free Batteries – Like many things in life, the term “maintenance-free” is only partially true. Lead-acid batteries normally consume some of the water in their dilute sulfuric acid electrolyte during a normal charge-discharge cycle. It actually electrolyzes into hydrogen and oxygen and escapes as gas. So adding water periodically is necessary to keep the plates flooded. Maintenance-free batteries use a calcium alloy of lead instead of an antimony alloy, which reduces the amount of electrolysis. In addition, the amount of free-standing electrolyte above the plates is designed to be much higher in a new maintenance-free battery. This means that there’s enough electrolyte to keep the plates covered even after a few seasons of normal use. So, during the battery’s normal service life there should be no need to add water. Any abnormal electrical system condition or high ambient temperatures may boil off more than the normal amount of water, however. Adding water may extend the service life of these supposedly maintenance-free batteries.
A maintenance-free battery, similar in design to a conventional automotive battery, is really just a heavier-duty version of the same arrangement. Many of the components have thicker construction, and different, more durable materials are typically used. For example, the plate grids often contain calcium, cadmium or strontium, to reduce gassing (which causes water loss) and self-discharge. This design is called a lead-calcium battery. The heavier-duty parts ensure that fluid loss is kept to a minimum and that components have a much longer life, making it a closed system.
Picking The Right Battery
Check your vehicle manual for the original equipment manufacturer’s recommendations for:
- Battery group size
- Cold cranking amps (CCA)
- Reserve capacity (RC) for your vehicle
Battery group size indicates the battery size that will best fit the physical dimensions of your vehicle. Many vehicles can accommodate more than one group size.
CCA is critical for good cranking ability. It’s the number of amps a battery can support for 30 seconds at a temperature of 0 degrees F until the battery voltage drops to unusable levels. A 12V battery with a rating of 600 CCA means the battery will provide 600 amps for 30 seconds at 0 degrees before the voltage falls to 7.20 V (six cells).
RC helps to power your vehicle’s electrical system if the alternator fails. It indicates the battery’s “staying power” — how many minutes the battery can supply ample power without falling below the minimum voltage needed to run your vehicle.
In general for both CCA and RC, the higher the number the better. HOWEVER, if you live in a cold climate, the CCA rating should be an important consideration in choosing a battery. Conversely, if you live in a high heat climate, you don’t need as much CCA.
If you’re looking for a deep cycle battery for marine or RV use, you must consider:
- The type of equipment to be powered
- The current (amps) needed to run the equipment
- The number of hours you’ll be using the equipment
Multiply the Amps by the Hours to determine the Amp Hours, or AH, required.
Look for a battery that will deliver the required amount of AH for the specified time and voltage. For a safety cushion, increase the number of AH by 20%. To add AH, connect batteries in parallel. To add voltage, connect batteries in a series.
Be careful of products that display only ratings such as Hot Cranking Amps (HCA) or Cranking Amps (CA)
Products that display HCA or CA ratings are tested at higher temperatures, in the case of HCA — 80 degrees — so the resulting numbers appear higher. (CAs are tested at 30°F.) Since these batteries are not as powerful as you might think, what looks like a “bargain” may end up costing you money. Make sure you are comparing apples to apples when looking at ratings. The CCA and RC are the best measure of a battery’s true power.
Check for freshness
Learn to interpret battery date codes. Or ask your retailer to make sure you purchase the “freshest” battery available. A battery that has been sitting on the shelf for extended periods can lose some of its charge and may not provide the performance you need during its first use. Long term performance probably won’t be compromised however, as the battery can be returned to its original levels of performance with either in-vehicle charging or by using an external charger.
Look for a hassle free warranty
- Is the warranty nationwide so you can obtain service wherever you are?
- How long is the free replacement period?
- After that time, will you be reimbursed for a portion of the battery’s cost on a prorated basis?
Consider value as well as price
You might want to get professional advice from your mechanic or retailer at your local parts store before you buy.
Purchase a new battery before your existing one fails.
Before you take a long trip, or when you’re having your car serviced, have your battery tested. It could save you a lot of time and money down the road.
Power Rating
Cold Cranking Amps: Cold Cranking Amps (CCA) is critical for good cranking ability. It refers to the number of amps a battery can support for 30 seconds at 0°F until the battery voltage drops to unusable levels. For example, a 12 volt battery with 600 CCAs means the battery will provide 600 amps for 30 seconds at 0°F before the voltage falls to 7.20 volts (six cells). The higher the CCA, the more powerful the cranking ability. As the temperature drops, the cranking power required by the car increases. However, as more cranking power is used, the amount of battery power available decreases.
If you live in a cold climate, you should consider the CCA rating when choosing a battery. If you live in a very hot climate, you don’t need as much CCA.
Reserve Capacity Rating: The second standard is called reserve capacity rating. This is a warm weather rating (80 degrees Fahrenheit), which estimates the amount of time it takes the terminal voltage of a fully charged battery to dip below 10.2 (or 1.7 volts per cell) at a continuous discharge rate of 25 amps. The rating is expressed in minutes. For example, a rating of 120 means the battery will run for two hours (120 minutes) before ceasing to function.
Battery Cycles (Charge & Discharge)
A battery has two main cycles, the charge and discharge cycles.
Discharge Cycle: In the discharge cycle, a chemical reaction takes place inside the battery in which the lead (Pb) of the negative plates combines with the SO4 of the sulfuric acid to produce lead sulfate (PbSO4). In this cycle, the electrolyte becomes weaker — specific gravity lessens — and the positive and negative plates become more like one another. Since the voltage, or charge, of a battery depends on the difference between the two plate materials and the concentration of the electrolyte, and since this difference decreases during discharging, the battery loses power. To anyone who has sat in a non-starting car and cranked away as the battery grew weaker and weaker, this scenario will be immediately recognizable.
Charge Cycle: In the charge cycle, the reverse is true. Electrical current, generated by the car’s alternator, passes through the plates, forcing SO4 back into the electrolyte bath and elevating specific gravity. Voltage increases.
Fluid Level
Formerly car batteries using lead-antimony plates would require regular watering top-up to replace water lost due to electrolysis on each charging cycle. By changing the alloying, more recent designs have lower water loss unless overcharged. Modern car batteries have reduced maintenance requirements, and may not provide caps for addition of water to the cells. If the battery has easily detachable caps then a distilled water top up may be required from time to time. Prolonged overcharging or charging at excessively high voltage causes some of the water in the electrolyte to be broken up into hydrogen and oxygen gases, which escape from the cells. If the electrolyte liquid level drops too low, the plates are exposed to air, lose capacity, and are damaged. The cells can be topped up with distilled or deionised water just above the visible plates. The sulfuric acid in the battery normally does not require replacement since it is not consumed even on overcharging.
Impurities in the water will reduce the life and performance of the battery. Manufacturers usually recommend use of demineralized or distilled water since even potable tap water can contain high levels of minerals.
Storing Batteries
When not in active use batteries need to be monitored and periodically charged to prevent damage and retain capacity. If you remember in the section above about battery types, typical starting batteries should not be allowed to drop under 90% charge. Make sure to fully charge the battery immediately prior to storage. Clean the battery with soap and water to remove any leakage paths on the battery case due to acid or sulphation buildup on the case exterior. Dry the unit and place in a cool (below 80F if possible), dry location environment. High temperatures increase the self discharge rate. Lead-acid batteries must always be kept in a fully charged condition. Check the battery voltage with a good digital volt meter every 6 months and charge it if it falls below 12.6 volts.
Remember, newer vehicles with on-board electronics such as computers, LCD screens, game systems, GPS units, clocks, etc., require battery power to retain system memory while the vehicle is parked. If the vehicle is to be stored for long periods you should use a maintenance charger to compensate for this drain. This charger should be voltage regulated between 13.2 – 13.8 volts, 1 amp maximum. On older vehicles, without electronics, disconnect the battery cables when the vehicle is not being used for extended periods.
Float chargers like these can be used when storing batteries or during cold weather. The floating circuit maintains a full charge without overcharging and it has an automatic safety shutoff.
Float Charger
If you’re leaving the vehicle parked outside but not driving it very often, you may also want to consider a solar charger.

Solar Charger
This solar charger simply plugs in to your cigarette lighter and compensates for small steady battery drains. Chargers like this can be found for under $20.00.
Changing A Battery
In most modern automobiles, the grounding is provided by connecting the body of the car to the negative electrode of the battery, a system called ‘negative ground’. The recommended practice when removing a car battery is to disconnect the ground connection first and then other terminal. This ensures that a short circuit will not occur by a wrench touching grounded engine parts while disconnecting the other terminal. Similarly, the ground should be connected last when installing a battery.
Mounting A Battery
It’s important that you properly mount the battery. Don’t hold the battery down with bungee or and leave it loose in a battery box. Loose hold-down straps or covers allow the battery to vibrate or bounce during vehicle operation. This can have several adverse effects. It can shake the active materials off the grid plates, severely shortening battery life. It can also loosen the plate connections to the plate strap, loosen cable connections, or even crack the battery case.

Above is a battery holder designed by Blue Torch Fab

Above is a battery holder for an Optima battery.

Here is a trunk mount battery kit. Don’t just buy one of these battery boxes from a parts store and put a battery in it. Make sure you by a bracket to hold the battery down. Note the red and black bracket and long bolts to hold the battery.
Battery Maintenance
With any type of car battery – sealed or unsealed – you must do the following to help keep it working properly:
1. Clean the cables. Disconnect the cables from the battery and clean them with a wire brush that has been doused in a home made mixture [the mixture combines one tablespoon of baking soda with one cup of water]. Use this same mixture to clean off the top of the battery; use a small and clean paint brush to apply the solution where needed. Don’t allow the solution to enter the cells of the battery.
2. Lubricate the posts. A small dab of petroleum jelly on each post will help keep your cables clean and free of corrosion longer. In addition, the jelly will make it easier for you to slip the cables back onto the battery. You can also use a corrosion preventative spray, then check the job by starting the car.
3. Check connections. Besides the connection between the cable and the posts, make sure that the battery hold down bar is securely in place. Not all cars have or need a battery hold down bar, but if yours has come so equipped, you will need to put it back in its proper place.
Battery maintenance is an easy task and one that should be performed on an annual basis. If you keep your battery properly maintained, you will extend its life and greatly reduce the chance that your battery will fail you at an inopportune time. Yes, the “maintenance free” label is incorrect, so please don’t be lulled into forgetting to do something that should never be overlooked.
Corrosion at the battery terminals can prevent a car from starting. To prevent corrosion, during regular battery service the terminals may be cleaned with a wire brush and corrosion products washed away with water. When the battery terminals are re-assembled, they are coated with Vaseline/petroleum jelly (grease is not desired) to reduce the rate of corrosion accumulation. The corrosion white powder is usually lead sulfate which is toxic by inhalation, ingestion and skin contact. It is also corrosive to the eyes and skin.
You can get an anti-corrosion spray for your battery terminals

You can get these felt rings to help prevent corrosion on the battery terminals
Battery Disposal
Never discard an old battery in the trash. Take the old battery to a recycling center that accepts automotive batteries. Generally, part stores charge a core charge when you purchase a new battery until you turn in your old battery to them.
Battery Defects

Common Battery Faults Include
- Shorted cell due to failure of the separator between the positive and negative plates
- Shorted cell or cells due to build up of shed plate material building up below the plates of the cell
- Broken internal connections due to corrosion
- Broken plates due to vibration and corrosion
- Low electrolyte
- Cracked or broken case
- Broken terminals
- Sulfation after prolonged disuse in a low or zero charged state
The primary wear-out mechanism is the shedding of active material from the battery plates, which accumulates at the bottom of the cells and which may eventually short-circuit the plates. If the battery shorts, it can get hot, boil and even explode.
Early automotive batteries could sometimes be repaired by dismantling and replacing damaged separators, plates, intercell connectors, and other repairs. Modern battery cases do not facilitate such repairs; an internal fault generally requires replacement of the entire unit.
Exploding Batteries
Any lead-acid battery system when overcharged will produce hydrogen gas. If the rate of overcharge is small, the vents of each cell allow the dissipation of the gas. However, on severe overcharge or if ventilation is inadequate or the battery is faulty, a flammable concentration of hydrogen may remain in the cell or in the battery enclosure. Any spark can cause a hydrogen and oxygen explosion, which will damage the battery and its surroundings and which will disperse acid into the surroundings. Anyone close to the battery may be severely injured. Potentially faulty batteries can often be detected by the swollen sides where pressure has risen owing to faulty valves.
Car batteries should always be handled with proper protective equipment (goggles, overalls, gloves).
In the photo above, the internal negative connection to the plates was poor by a manufacturing defect and when the rather high starter motor current flowed, the connection failed and there was a spark inside the battery that triggered the explosion.
The battery above was being used with a generator. When the generator went to start, a loud boom was heard. It appears that when the generator went to start, a spark in the battery ignited hydrogen gas which cause it to explode.
The explosion doesn’t necessarily have to be as dramatic as the other batteries shown. You can see where the battery above is cracked at the front top corners and a piece is blown out of the top.
Frequently Asked Questions (FAQ’s)
Q – What should I consider when buying a battery?
A- SIZE: What are the dimensions of your original battery? POWER: What are the Cold Cranking Amps required to power your vehicle? WARRANTY: Automotive batteries are backed by a warranty package. Chose what is right for your vehicle’s needs.
Q – Why is it important to remove the ground wire first?
A- Before you start, always check the type of grounding system the vehicle has. If you remove the positive connector first in a negative ground system, you risk the chance of creating a spark. That could happen if the metal tool you’re using to remove the positive terminal connector comes in contact with any piece of metal on the car. If you are working near the battery when this occurs, it might create an ignition source that could cause the battery to explode. It’s extremely important to remove the ground source first.
Q- How can I tell how fresh a battery is?
A – You can usually find a small decal on the side of the battery container giving you the month and year the battery was shipped out of the plant. The letter corresponds with the month, starting with “A” for January, “B” for February, and so on. The number represents the year with “9” standing for 1999, “0” for 2000, and so on. A9, would be January, 1999. C0 would be March, 2000.( The letter “I” is skipped so the letter “M” would be December.)
Q- What does CCA mean?
A – Cold Cranking Amps is a rating used in the battery industry to define a battery’s ability to start an engine in cold temperatures. The rating is the number of amps a new, fully charged battery can deliver at 0° Farenheit for 30 seconds, while maintaining a voltage of at least 7.2 volts, for a 12 volt battery. The higher the CCA rating, the greater the starting power of the battery.
Q – What are MCA or CA rates?
A – This is a rating used to describe the discharge load in amperes which a new, fully charged battery at 32 degrees F (0C), can continuously deliver for 30 seconds and maintain a terminal voltage equal or greater than 1.2 volts per cell. It is sometimes referred to as Marine Cranking Amps or Cranking Amps.
Q – What is Reserve Capacity?
A – Reserve Capacity, (RC) is a battery industry rating, defining a battery’s ability to power a vehicle with an inoperative alternator or fan belt. The rating is the number of minutes a battery at 80 degrees F can be discharged at 25 amps and maintain a voltage of 10.5 volts for a 12 volt battery. The higher the reserve rating, the longer your vehicle can operate should your alternator or fan belt fail.
Q – What can excessive heat do to batteries?
A – Hot temperatures will deteriorate a battery’s life quicker by evaporating the water from the electrolyte, and corroding and weakening the positive grids.
Q – What is causing my battery to get hot and boil?
A – Boiling can be caused by either overcharging or from it internally shorting from one of the plates getting loose and touching another plate. Be careful because a shorting and or boiling battery can explode.
Q – My engine was running for a while and I was showing 14.2 volts at the battery terminals. A while after shutting the vehicle off, I checked battery voltage and it was only 10.3 volts. Why?
A- A battery is considered charged at 12.4 volts. Each cell has 2.1 volts. If one cell isn’t working, your 12.4 volt charged battery is only going to be capable of 10.3 volts (12.1 volts – 2.1 volts = 10.3 volts)
Terms
Ampere-hours (A·h) is the product of the time that a battery can deliver a certain amount of current (in hours) times that current (in amps), for a particular discharge period. This is one indication of the total amount of charge a battery is able to store and deliver at its rated voltage. This rating is rarely stated for automotive batteries.
Cranking amps (CA), also sometimes referred to as marine cranking amps (MCA), is the amount of current a battery can provide at 32°F (0°C). The rating is defined as the number of amperes a lead-acid battery at that temperature can deliver for 30 seconds and maintain at least 1.2 volts per cell (7.2 volts for a 12 volt battery).
Cold cranking amps (CCA) is the amount of current a battery can provide at 0°F (−18°C). The rating is defined as the current a lead-acid battery at that temperature can deliver for 30 seconds and maintain at least 1.2 volts per cell (7.2 volts for a 12-volt battery). It is a more demanding test than those at higher temperatures.
Hot cranking amps (HCA) is the amount of current a battery can provide at 80°F (26.7°C). The rating is defined as the current a lead-acid battery at that temperature can deliver for 30 seconds and maintain at least 1.2 volts per cell (7.2 volts for a 12-volt battery).
Reserve capacity minutes (RCM), also referred to as reserve capacity (RC), is a battery’s ability to sustain a minimum stated electrical load; it is defined as the time (in minutes) that a lead-acid battery at 80°F (27°C) will continuously deliver 25 amperes before its voltage drops below 10.5 volts.
Battery Council International group size (BCI) specifies a battery’s physical dimensions, such as length, width, and height. These groups determined are by the Battery Council International organization.
SLI battery (Starting – Lighting – Ignition) is a typical battery used to power the starter motor, the lights, and the ignition system of a vehicle’s engine.
Peukert’s Law expresses the fact that the capacity available from a battery varies according to how rapidly it is discharged. A battery discharged at high rate will give fewer ampere hours than one discharged more slowly.
1983-2011 Ford Ranger Battery Group Sizes
The Ford Ranger used several different battery group sizes from 1983-2011 depending on engine size, electrical package, cold weather packages, and model year.
| Years | Typical OEM Battery Group | Alternate / Optional Groups | What Determined It |
|---|---|---|---|
| 1983-1988 | Group 58 | Group 65 (some V6 / heavy-duty applications) | Engine size, HD electrical package |
| 1989-1996 | Group 58 or 65 | — | 4-cylinder vs V6, tray size |
| 1997-2011 | Group 59 | Group 58 (some applications) | Mostly standardized tray/cable layout |
| 1998-2011 (some aftermarket catalogs) | Group 59 or 65 | Group 58 | Modern replacement supersessions |
What Determined Which Battery a Ranger Got?
1. Engine Size
This was the biggest determining factor.
- 2.0L / 2.3L / 2.5L Four-Cylinders
- Usually Group 58
- Lower CCA requirement
- Smaller tray common
- 2.8L / 2.9L / 3.0L / 4.0L V6
- Often Group 65
- Higher cranking requirement
- Larger tray or hold-down
- 1997-2011 Rangers
- Ford largely standardized around Group 59
2. Heavy Duty Electrical / Tow Packages
Some Rangers equipped with towing packages, air conditioning, cold-weather packages, or higher-output alternators received larger batteries, usually Group 65.
3. Battery Tray and Hold-Down
The tray itself often determines what fits better than modern parts catalogs. Many Rangers can physically accept Groups 58, 59, and 65 with little or no modification.
1984-1990 Ford Bronco II Battery Group Sizes
The Bronco II shared most of its drivetrain and electrical systems with the Ranger, so battery sizing was very similar.
| Years | Engine | Typical OEM Battery |
|---|---|---|
| 1984-1985 | 2.8L V6 | Group 58 or Group 65 |
| 1986-1990 | 2.9L V6 | Group 65 most common |
| 1986-1990 | 2.3L Diesel (rare) | Group 65 heavy-duty |
The Bronco II often used the larger Group 65 because:
- Most models were V6-powered
- Many were equipped with 4WD
- The Bronco II was heavier than comparable Rangers
- Electrical demand was typically higher
What Battery Fits Best in a Bronco II?
| Battery Group | Fits Stock Tray? | Notes |
|---|---|---|
| Group 56 | Yes | Modern catalog replacement |
| Group 58 | Yes | Likely closest to original fitment |
| Group 65 | Usually | Often fits with minor hold-down adjustment |
| Group 34/78 | Yes | Excellent AGM upgrade |
| Group 24F | Sometimes | Requires careful measuring |
Why Modern Parts Stores List Group 56 Batteries
Many modern parts catalogs now list a Group 56 battery for older Ford applications including the Bronco II.
That does not necessarily mean the vehicle originally came with a Group 56 battery from the factory.
What happened is:
- Older Ford compact trucks commonly used Group 58 batteries
- Group 58 batteries became less common in retail inventory
- Parts stores substituted Group 56 because it physically fits many applications
- Modern catalogs standardized around Group 56 as a replacement size
So while Group 56 is commonly listed today, many enthusiasts and longtime Ford technicians still recognize Group 58 as the more original-style battery for many 1980s Rangers and Bronco IIs.
Physical Battery Size Comparison
| Battery Group | Length | Width | Height | Typical Use |
|---|---|---|---|---|
| Group 56 | 10.9″ | 6.9″ | 8.4″ | Modern replacement for older Ford compact trucks |
| Group 58 | 10.1″ | 7.2″ | 6.9″ | Common original Ranger / Bronco II battery |
| Group 59 | 10.1″ | 7.6″ | 7.75″ | Common in 1997-2011 Rangers |
| Group 65 | 12.1″ | 7.5″ | 7.6″ | Higher-capacity V6 / HD battery |
| Group 34 | 10.2″ | 6.8″ | 7.9″ | Popular AGM upgrade |
| Group 34/78 | 10.2″ | 6.8″ | 7.9″ | Optima-style dual terminal AGM battery |
| Group 24F | 10.8″ | 6.8″ | 8.9″ | Possible swap with tray measurements |
| Group 75 | 9.1″ | 7.1″ | 7.9″ | Possible fit depending on terminal orientation |
Best Battery for Overlanding
Whether you’re building a 1983-2011 Ford Ranger or a 1984-1990 Ford Bronco II for overlanding, the battery becomes far more important than it is in a normal daily driver.
Overland vehicles often spend extended periods running accessories while the engine is off, including:
- Camping lights
- Portable refrigerators
- Radios and communication equipment
- Air compressors
- USB charging systems
- GPS and navigation equipment
- Winches
- Inverters
Because of this, the best overlanding battery should prioritize:
- Reserve Capacity (RC)
- Cold Cranking Amps (CCA)
- Vibration resistance
- Deep-cycle tolerance
- Durability in extreme temperatures
- Reliability in remote areas
Best Overall Choice: Group 65 AGM Battery
For most Ranger and Bronco II overland builds, a Group 65 AGM battery is the best overall solution.
Compared to smaller Group 56, 58, or 59 batteries, the Group 65 provides:
- More reserve power for accessories
- Higher cold cranking performance
- Longer runtime with the engine off
- Better resistance to vibration and trail abuse
- Improved recovery after repeated discharge cycles
- Better support for future upgrades like winches and onboard air
AGM batteries are especially useful in off-road vehicles because they are sealed and spill-proof. Unlike traditional flooded batteries, AGM batteries tolerate vibration, rough terrain, and off-camber angles much better.
Modern Group 65 AGM batteries commonly offer:
- 750-950 CCA
- 120-150 minutes reserve capacity
- 70-75 amp-hour capacity
That makes them ideal for vehicles with heavier accessory loads and extended backcountry travel. ODYSSEY Performance Series Group 65 AGM batteries and similar batteries commonly fall into this range.
Why AGM Batteries Work Well for Overlanding
| Feature | Benefit for Overlanding |
|---|---|
| Sealed Construction | No acid spills on rough trails |
| Vibration Resistance | Better durability off-road |
| Deep-Cycle Capability | Handles repeated accessory use better |
| Low Self-Discharge | Better for vehicles sitting during trips |
| Higher Reserve Capacity | Longer runtime for refrigerators and lighting |
| Faster Recovery | Recharges more efficiently after heavy use |
Many overlanders and off-road enthusiasts prefer AGM batteries because they provide a balance between starting performance and deep-cycle capability. Discussions within the overlanding community regularly highlight the long-term durability and reserve capacity advantages of AGM batteries in expedition-style vehicles.
Recommended Battery Sizes for Overlanding
| Vehicle | Recommended Overland Battery | Notes |
|---|---|---|
| 1983-1988 Ford Ranger | Group 65 AGM | May require minor hold-down adjustment |
| 1989-1996 Ford Ranger | Group 65 AGM | Usually fits easily in V6 trucks |
| 1997-2011 Ford Ranger | Group 65 AGM or Group 59 AGM | Depends on tray size and cable reach |
| 1984-1990 Ford Bronco II | Group 65 AGM | Best overall balance of capacity and fitment |
Popular AGM Battery Choices
- Optima YellowTop D65
- Odyssey Extreme Series Group 65
- Duralast Platinum AGM Group 65
- XS Power D6500
- Interstate AGM Group 65
Premium AGM batteries such as the Odyssey Extreme Series offer up to 950 CCA and approximately 145 minutes of reserve capacity, making them excellent choices for heavily-equipped overland rigs.
Budget-Friendly Overlanding Option
If cost or remote serviceability is more important than maximum performance, a traditional flooded Group 65 battery is still a very solid option.
Advantages include:
- Lower replacement cost
- Easier to find in small towns
- Simpler charging requirements
- Good reliability for moderate accessory loads
For many casual overland builds, a high-quality flooded Group 65 battery combined with upgraded battery cables and a healthy charging system is more than adequate.
Important Charging System Considerations
Before installing a large AGM battery, it is important to make sure the charging system is healthy.
Older Rangers and Bronco IIs often benefit from:
- Upgraded battery cables
- Improved engine and chassis grounds
- A higher-output alternator
- Clean battery terminals
- Modern voltage regulation
Some AGM owners have reported charging issues when using weak or aging alternators that cannot properly recharge AGM batteries after heavy accessory use.
Dual Battery Systems
For serious overlanding builds running refrigerators, solar systems, inverters, onboard air, communications equipment, or winches, a dual battery setup may be the best long-term solution.
A common overland setup includes:
- Main AGM starting battery
- Secondary auxiliary battery
- Battery isolator or DC-DC charger
- Solar charging capability
This allows camping accessories to run from the auxiliary battery while preserving enough power to reliably start the vehicle.
AGM vs LiFePO4 Auxiliary Batteries
Many modern overland builds are now using Lithium LiFePO4 batteries as the auxiliary battery instead of a traditional AGM deep-cycle battery.
LiFePO4 batteries offer several advantages:
- Much lighter weight
- Higher usable capacity
- Faster charging
- Longer cycle life
- Better efficiency
However, they also have some important limitations that Ranger and Bronco II owners should consider.
Many LiFePO4 batteries are not designed for engine starting duties and should not replace the main starting battery unless specifically designed as a starting battery.
Heat is another important consideration. Unlike AGM batteries, many LiFePO4 batteries do not tolerate prolonged under-hood heat very well. Because of this, many overlanders mount LiFePO4 auxiliary batteries:
- Inside the cab
- In the cargo area
- Inside a storage drawer system
- In a ventilated camper or trailer compartment
This is especially important on older Rangers and Bronco IIs where under-hood temperatures can become very high during slow-speed trail driving or summer travel.
Most LiFePO4 auxiliary battery setups also require a proper DC-DC charger rather than a simple battery isolator. This helps ensure the lithium battery receives the correct charging profile and prevents charging-system issues.
For many overland builds, the best overall setup is often:
- Main Battery: Group 65 AGM starting battery
- Auxiliary Battery: LiFePO4 house battery
This combination provides reliable cold-weather engine starting while still taking advantage of lithium battery capacity and efficiency for camping accessories.
For a more detailed explanation of lithium battery technology, charging requirements, and safety considerations, see our Lithium Battery Basics article.
Final Recommendation
| Build Type | Recommended Battery |
|---|---|
| Stock Restoration | Group 58 |
| Daily Driver | Group 56 or Group 58 |
| Mild Overland Build | Group 65 Flooded or AGM |
| Serious Overland Build | Group 65 AGM |
| Expedition / Heavy Accessories | Dual AGM Battery System |
For most 1983-2011 Ford Rangers and 1984-1990 Ford Bronco IIs, the Group 65 AGM is the best balance of:
- Capacity
- Reliability
- Accessory support
- Trail durability
- Cold-weather performance
Before purchasing any battery, always verify:
- Battery tray dimensions
- Hold-down location
- Hood clearance
- Cable reach
- Terminal orientation
Many Rangers and Bronco IIs have had trays, cables, and battery hold-downs changed over the past 40 years, so physical measurements are often more reliable than modern parts catalogs alone.
Related Articles
Adding An Auxiliary Battery To A 2019+ Newer Ford Ranger
Last Updated: April 16, 2026
About The Author
Jim Oaks is the founder of TheRangerStation.com, the longest-running Ford Ranger resource online since 1999. With over 25 years of hands-on experience building and modifying Ford Rangers — including magazine-featured builds like Project Transformer — Jim has become one of the most trusted authorities in the Ford Ranger off-road and enthusiast space.
Since launching TheRangerStation.com, Jim has documented thousands of real-world Ranger builds, technical repairs, drivetrain swaps, suspension modifications, and off-road adventures contributed by owners worldwide. TheRangerStation.com has been referenced in print, video and online by enthusiasts, mechanics, and off-road builders looking for practical, and experience-based information.
