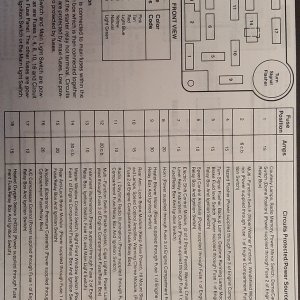
It takes exactly as much oxygen to burn the hydrogen as you made from the water. Maybe a little more if it leaks. This is one of many reasons why it can't possibly work. There is NO chance there is any left to burn gasoline AND all the hydrogen. If you're not burning all the hydrogen, it's a (bad) oxygen generator that displaces fuel with hydrogen. If you ARE burning the hydrogen, you didn't do anything.
And it ain't pure oxygen.
And I can make an airtight seal over a tube with my hand. You can't?
I'm thoroughly amazed at how badly one has to understand the most basic chemistry in order to think something like this might have the slightest prayer.
Now this statement smacks of someone who's too big for his britches

And tells me you've never tried any of this stuff to prove yourself wrong. It's akin to someone from the 1800's saying man will never be able to travel to the moon.

If it took one part oxygen to burn two parts hydrogen, then water would be flamable.

And why wouldn't the oxygen produced not be pure?

 Did you ever stop to think about what the two components of water are? You don't need air
Did you ever stop to think about what the two components of water are? You don't need air 
 If it took one part oxygen to burn two parts hydrogen, then water would be flamable.
If it took one part oxygen to burn two parts hydrogen, then water would be flamable.  And why wouldn't the oxygen produced not be pure?
And why wouldn't the oxygen produced not be pure?  I'm sure if I had the time to look em up, there are thousands of things invented by us Rednecks,(of which throughout the 1800's and early 1900's there really were hundreds of uneducated inventors who could be classified by you as Rednecks) of which their inventions were likely stolen by intellectuals(like yourself) as your own. And it's really hard to pay attention to your grammar. It really doesn't make sense to us common folk.
I'm sure if I had the time to look em up, there are thousands of things invented by us Rednecks,(of which throughout the 1800's and early 1900's there really were hundreds of uneducated inventors who could be classified by you as Rednecks) of which their inventions were likely stolen by intellectuals(like yourself) as your own. And it's really hard to pay attention to your grammar. It really doesn't make sense to us common folk.